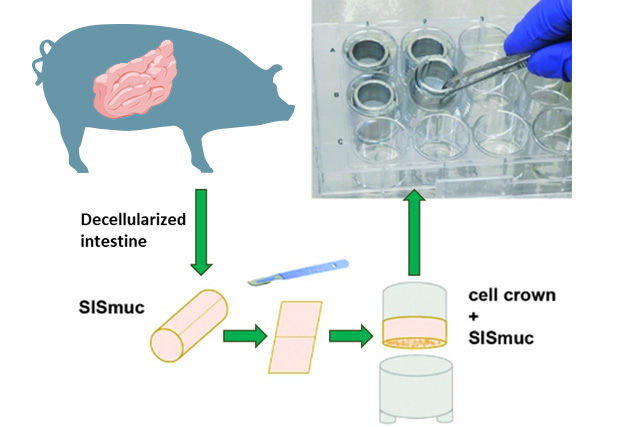
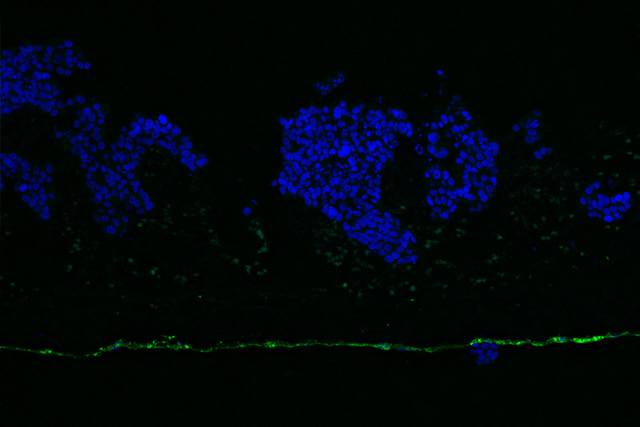
Our portfolio

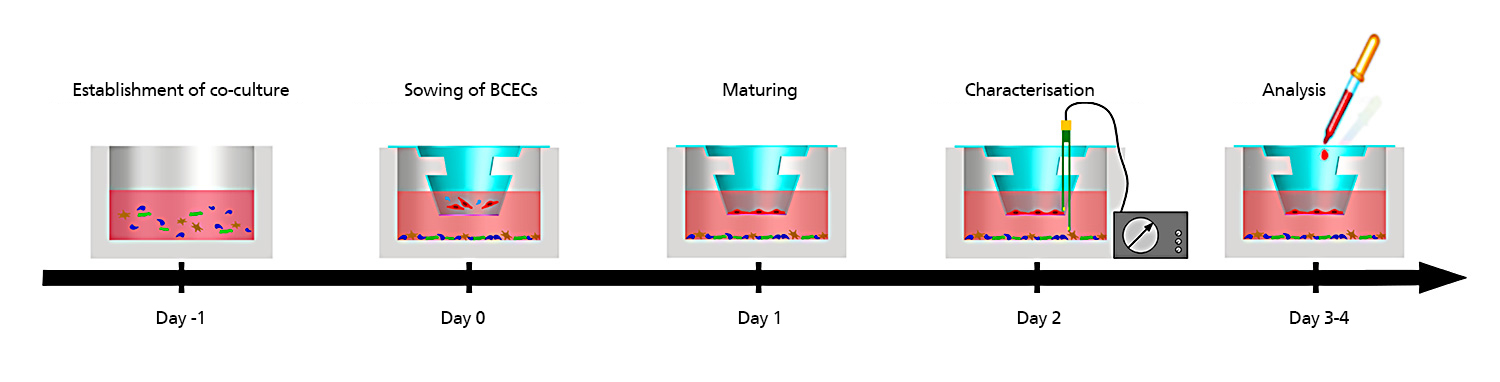
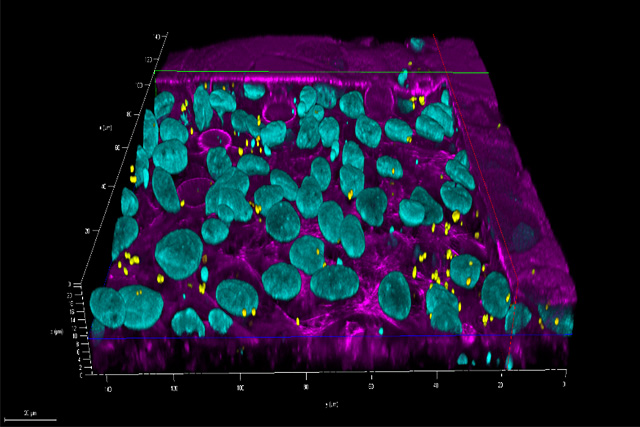
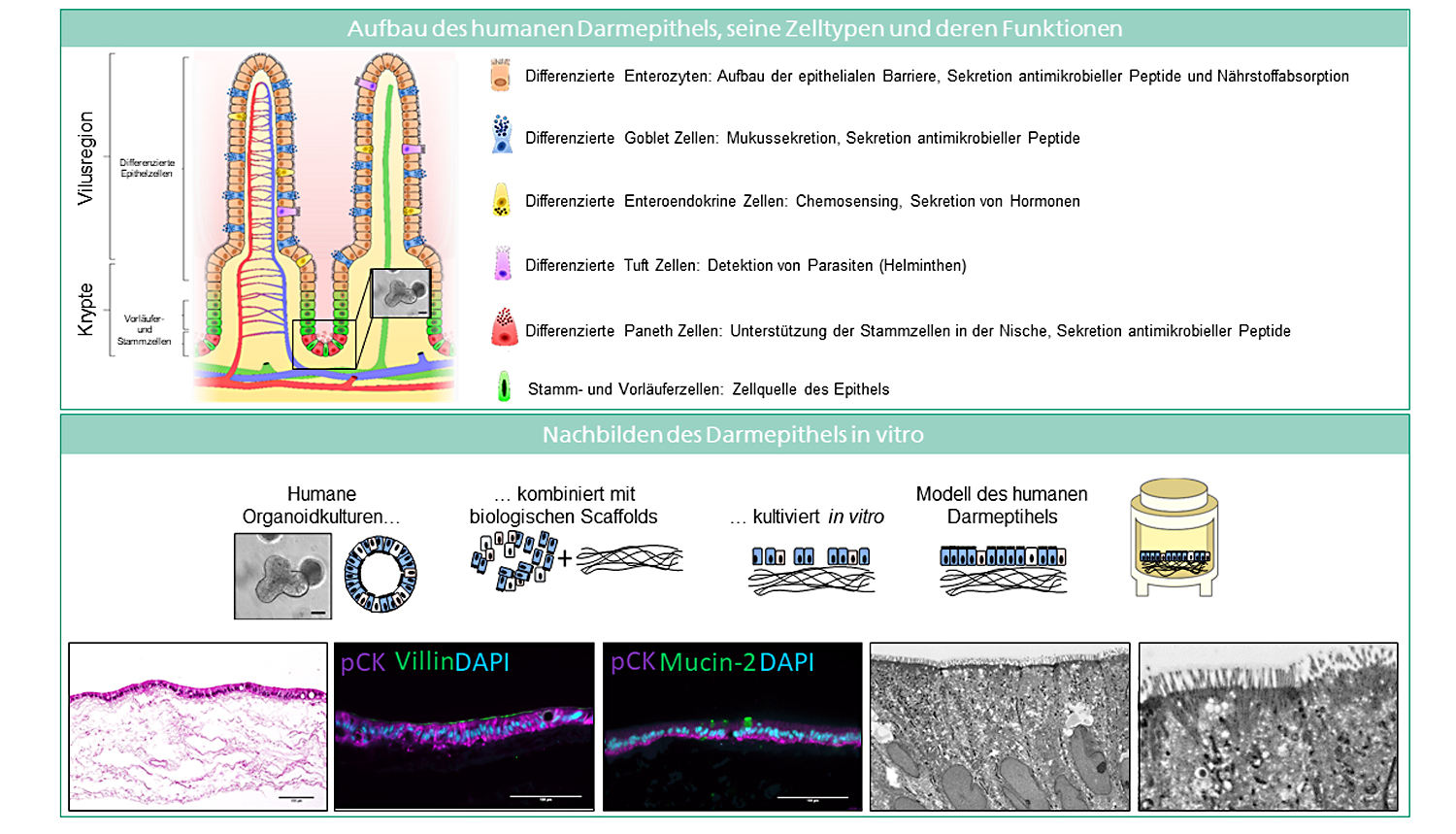
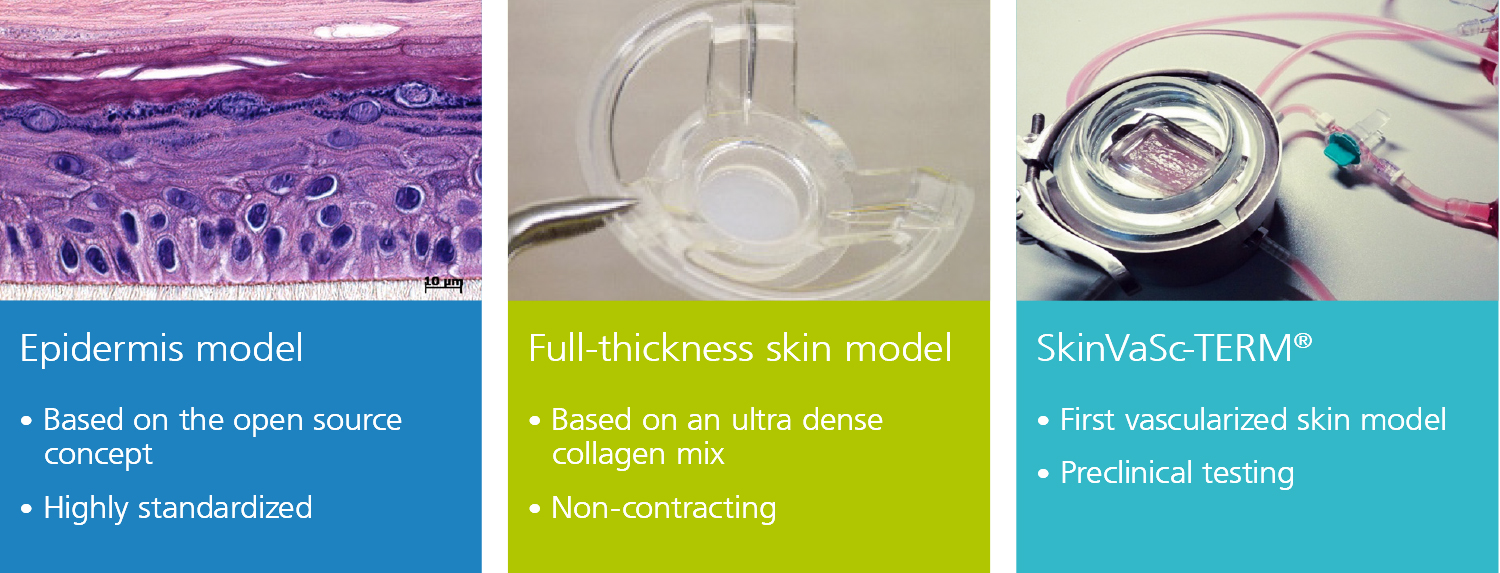
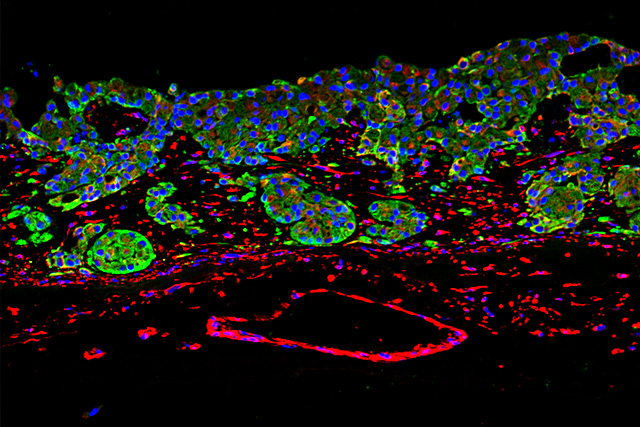
At the Fraunhofer TLC-RT, there is a broad portfolio of in vitro tissue models available for a wide variety of questions. Within our portfolio, we focus on barrier organs such as the skin, intestine, respiratory tract, or the blood-brain barrier. The models are of human origin and are generated either from primary cells or stem cells such as induced pluripotent stem cells. The aim is to make an active contribution to the reduction of animal experiments without neglecting the necessary efficacy testing or risk assessment. Hence, the TLC-RT commits itself to a high standardization of the models and industrial applicability to bridge the gap between academic and industrial research.
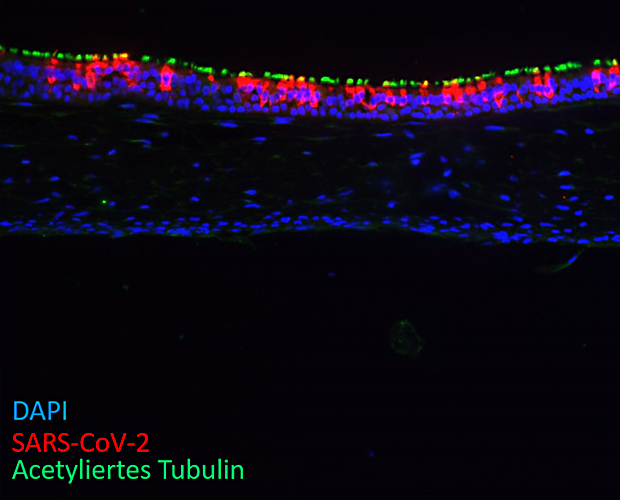
Besides the healthy state, we can also mimic diseased tissues to determine the efficacy of novel therapeutics in a fully human setting. Due to the high predictive capacity of the test systems, the risk of cost-intensive failures in the translation from the preclinical to the clinical phases can be minimized.